31 July 2017
Constipation is one of the most common non-motor related complaints affecting Parkinson’s disease (PD) patients. Two important studies from the same research group published in the Journal of Parkinson’s Disease expand the understanding of the relationship between PD and gastrointestinal dysfunction. In one study, investigators measured actual colonic dysfunction and compared it to reported constipation. In the other study, researchers tracked the position of an ingested wireless electromagnetic capsule using the novel 3D-Transit system in order to calculate gastrointestinal (GI) regional transit times.
The reliability of patient reporting can vary from person-to-person, especially when it comes to conditions with flexible criteria or symptoms like constipation. Researchers wanted to gauge how actual colonic function compared to subjective constipation reports. In their study, “Objective Colonic Dysfunction Is Far More Prevalent than Subjective Constipation in Parkinson’s Disease: A Colon Transit and Volume Study,” researchers tracked colonic transit time (CTT) using radio opaque markers and colonic volume in PD patients traced via CT scan. They found significantly delayed CTT and increased volume in PD patients.
“In the present study, 79% of early-stage PD patients had significantly increased CTT and 66% showed increased colonic volume,” said lead investigator Per Borghammer, MD, PhD, DMSc, Department of Clinical Medicine – Positron Emission Tomography Center, Aarhus University, Aarhus, Denmark. “The difference in CTT was more pronounced in the transverse, descending, and rectosigmoid segments of the colon, indicating that early stage PD patients exhibit a combination of slow transit and outlet constipation.”
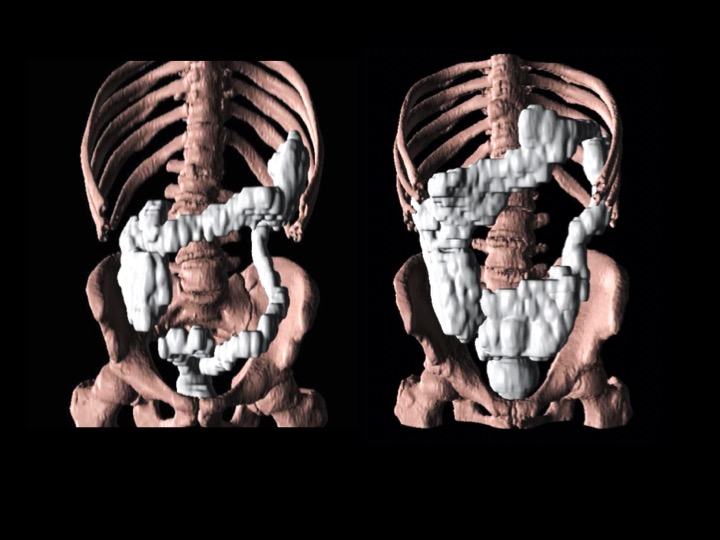
Colonic volume in a healthy control subject (left) and a PD patient with severely increased volume (right).
Gastric emptying and completeness of medication absorption was also studied. Investigators found no difference in the rate of gastric emptying between the control group and patients with PD. As for malabsorption, while previous observations have suggested that PD patients may have problems absorbing tablets in the intestinal tract, in this study, non-absorbed tablets were only seen in two PD subjects and no residues were found in the stomach. As Dr. Borghammer explained, “These negative findings go against the commonly held notion that slowed gastric emptying and malabsorption of medication is a common problem in early stage PD. However, at later disease stages it may be another matter.”
In addition, the team collected patient feedback about their upper GI symptoms and constipation using the Rome III criteria, NMSQuest, and the Cleveland constipation score. Objective colonic dysfunction was considerably more common than self-reported constipation, which investigators attributed to the use of different questionnaires. “These findings highlight the need for more research on how to define constipation in PD and also the need for improved understanding of the relationship between subjective symptoms and objective dysfunction,” added Dr. Borghammer.
Trying to define constipation can be complicated and as the first study showed, can vary according to patients’ perceptions; however, in the second study, investigators used the latest technology to collect objective data on GI transit times in PD patients, minimizing the risk for misinterpretation. In “Gastrointestinal Transit Time in Parkinson’s Disease Using a Magnetic Tracking System”, the team used the 3D-Transit system to observe motility in both the upper and lower GI tract. The system tracks the position of an ingested wireless electromagnetic capsule to more accurately measure segmental transit times in the GI canal.
Results showed that transit time and colonic motility patterns were significantly affected in PD patients. “Using the ambulatory 3D-Transit system we report exact small intestinal transit time (SITT) data in PD for the first time. The SITT was significantly increased compared to control subjects. We also demonstrated a highly significant increase in proximal colonic transit time, and a decrease in colonic mass- and fast movements in the PD patients,” said Dr. Borghammer.
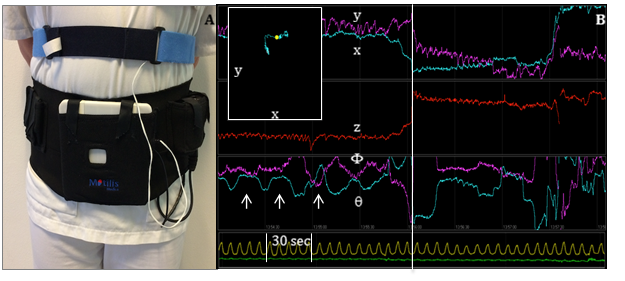
3D-Transit system. A. Sensors in the detector plate register electromagnetic signals from the ingested capsule. A chest worn respiration belt registers artifacts due to respiration. B. Graphs displaying position (x, y, z) and orientation (F, q) of the capsule when passing the GI tract. Upper left window shows capsule position in x-y direction according to the external monitor. Arrows mark the contraction frequency of 3 per minute, characteristic of the stomach. Vertical line marks the shift in capsule position from stomach to small intestine. Yellow line marks respiration. Green line marks accelerometer.
By using these markers to track GI function over a 24-hour period, researchers were able to gather crucial data about the bowel of PD patients. The information revealed that dysfunction was more widespread than previously known, with the trackers showing that delayed transit times affected not only the colon, but also the small intestine. This dysfunction appears to gradually increase from the proximal to the distal part of the GI tract.
The gut plays an important role in PD. New research, like these two studies, is essential to unlocking a better understanding of how the disease affects GI function. “Our studies suggest that gut problems are markedly under-diagnosed when relying solely on the subjective symptoms of the patients. In short, about 40% of PD patients complain of constipation but up to 80% have objective dysfunction of the colon.”
# # #
NOTES FOR EDITORS
“Objective Colonic Dysfunction Is Far More Prevalent than Subjective Constipation in Parkinson’s Disease: A Colon Transit and Volume Study,” by Karoline Knudsen, Tatyana D Fedorova, Anne C Bekker, Peter Iversen, Karen Østergaard, Klaus Krogh and Per Borghammer (DOI 10.3233/JPD-161050) and “Gastrointestinal Transit Time in Parkinson’s Disease Using a Magnetic Tracking System,” by Karoline Knudsen, Anne-Mette Haase, Tatyana D. Fedorova, Anne Charlotte Bekker, Karen Østergaard, Klaus Krogh, and Per Borghammer (DOI 10.3233/JPD-171131), published in the Journal of Parkinson’s Disease, Volume 7, Issue 2, by IOS Press.
For additional information contact Diana Murray, IOS Press, at +1 718 640-5678, d.murray@iospress.com. Journalists wishing to interview the studies’ authors should contact Karoline Knudsen at karoline.knudsen@aarhus.rm.dk.
ABOUT IOS PRESS
Celebrating its 30th anniversary, IOS Press (www.iospress.com) is headquartered in Amsterdam with satellite offices in the USA, Germany, India and China and serves the information needs of scientific and medical communities worldwide. IOS Press now publishes over 100 international journals and about 75 book titles each year on subjects ranging from computer sciences and mathematics to medicine and the natural sciences.







